Proudly part of the City Fertility Network
Proudly part of the City Fertility Network
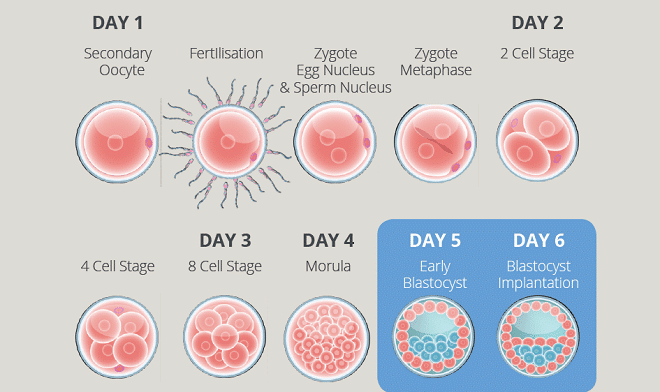
A culture media system is used in the laboratory to optimise embryo development in vitro (in glass) by supporting metabolic and physiological phases before implantation. Improvement of embryo culture has contributed significantly to the increase in the overall success rates in assisted reproductive technology (ART).
EmbryoGen® and BlastGen™ are new sequential culture media offered to City Fertility patients. Fertilised and cleavage-stage embryos (zygote to day three) are cultured with EmbryoGen®, followed by the culture to the blastocyst stage with BlastGen™. For the embryos cultured with this system, BlastGen™ is the recommended medium for embryo transfer.
EmbryoGen® and BlastGen™ are suitable for all patients and are especially recommended for people who have experienced recurrent biochemical pregnancy loss, miscarriage, recurrent implantation failures or unexplained infertility. EmbryoGen® and BlastGen™ are culture media that have been developed with the addition of the cytokine Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF).
GM-CSF is the active ingredient in EmbryoGen® and BlastGen™. A protein expressed in the female reproductive tract, GM-CSF is upregulated throughout embryo development. Research has shown that an addition of 2ng/ml of GM-CSF to culture media improved embryo growth and development.
GM-CSF appears to assist embryo development in several ways:
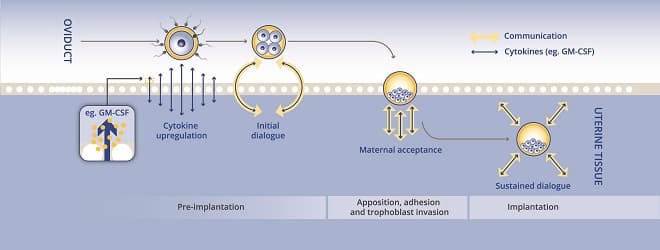
A large clinical trial conducted in Denmark and Sweden observed the effects of GM-CSF and its addition in culture media of patients undergoing ART. Studies have shown a significant increase in survival of transferred embryos and lower miscarriage rates. GM-CSF displays positive effects on embryo development and there are no known downsides of culturing embryos in culture media containing 2ng/ml of GM-CSF.
EmbryoGen® has CE (European), FDA (American Food and Drug Administration) and Canada Medical Device approval. BlastGen™ is in the process of being CE-marked and FDA-cleared, and has been approved by Health Canada. In Australia, Therapeutic Goods Administration (TGA) approval is pending. Patients of City Fertility can access both EmbryoGen® and BlastGen™ through the TGA’s Authorised Prescriber Scheme.
If you would like to discuss the use of EmbryoGen® and BlastGen™ in your treatment cycle, please consult your treating specialist or contact the City Fertility scientists.
To learn more, please read our fact sheet: EmbryoGen and BlastGen and our blog article: Breakthrough IVF technology BlastGen and EmbryoGen at City Fertility.
Request an appointment with one of our specialists.Make your appointment