What is ICSI?
Intracytoplasmic Sperm Injection (ICSI) is a specialised form of In Vitro Fertilisation (IVF) that is used for the treatment of severe cases of male factor infertility. ICSI is usually recommended where there is low sperm count, a high number of abnormally shaped sperm, reduced sperm motility, no sperm in a semen analysis or following an unsuccessful vasectomy reversal. It is also recommended for couples who have had previous cycles where standard IVF has resulted in reduced or no fertilisation.
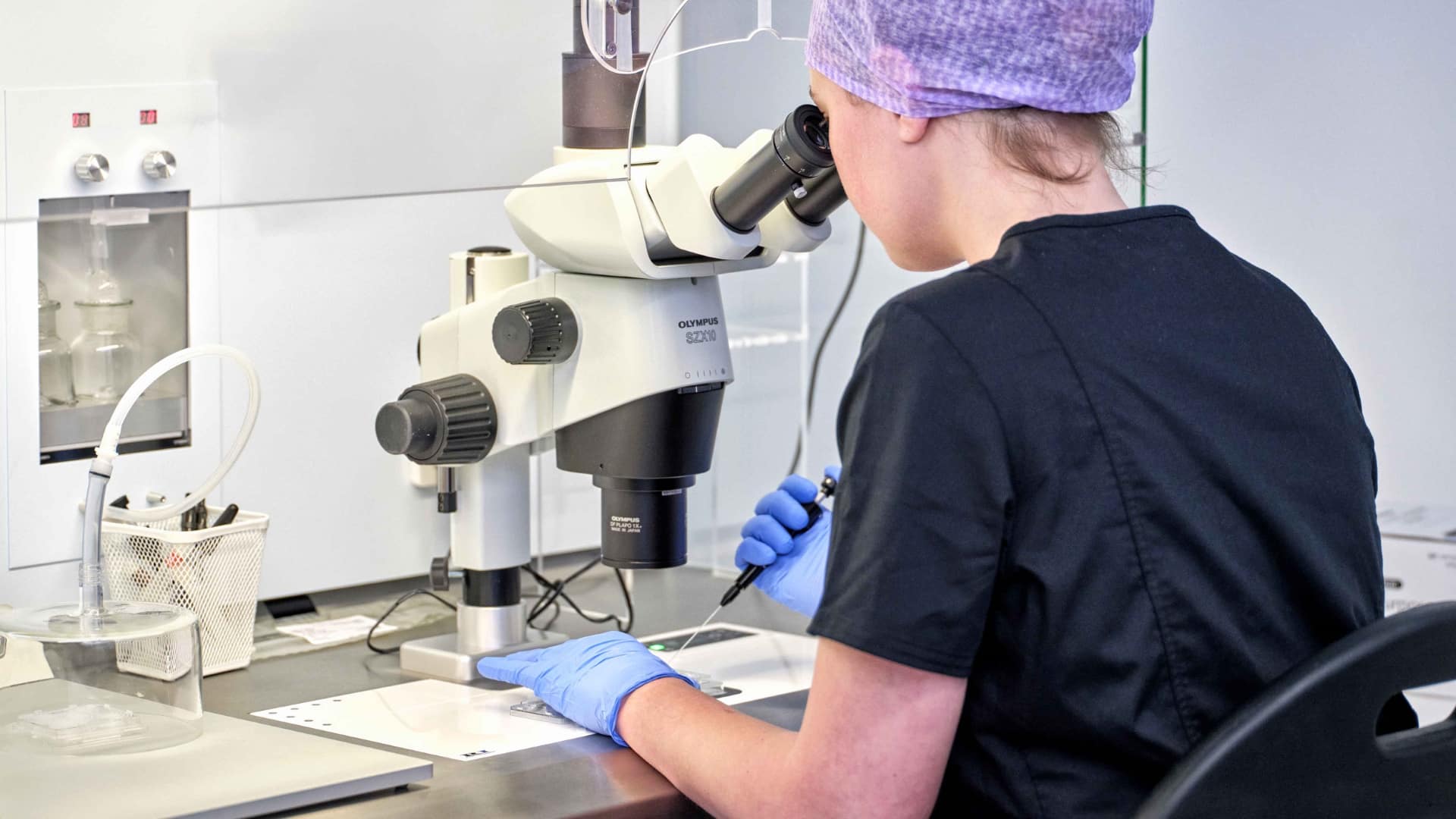
The process of ICSI involves the injection of a single sperm directly into a mature egg by a skilled embryologist.
This differs from standard IVF, where multiple sperm are added to the eggs in a special dish and left to fertilise without intervention from an embryologist.
Request an appointment with one of our specialists.Book an Initial Consultation
What does the ICSI process look like?
The purpose of ICSI is to improve the chances of fertilisation in cases where standard IVF may not be successful. The process involves the selection of a single swimming sperm for injection into a single mature egg. Every mature oocyte (egg) retrieved is injected and then assessed for fertilisation 16-18 hours post-injection.
The first step of an ICSI IVF cycle is ovarian stimulation. It involves a series of daily follicle-stimulating hormone (FSH) injections self-administered at home from Day 2 or 3 of your menstrual cycle. This medication is used to stimulate the ovaries to encourage the growth of multiple follicles containing eggs.
Your fertility specialist will closely monitor your ovarian response during the stimulation phase with ultrasounds and/or blood tests. This is important as medications may be adjusted to control the size and the number of follicles.
To assist with the final maturation of the egg and loosening of the egg from the follicle wall ready for egg collection, an injection of Human Chorionic Gonadotrophin (hCG). This is often referred to as ‘the trigger injection’.
The egg retrieval is performed 35-38 hours after ‘the trigger injection’ under ultrasound guidance and takes place while you’re lightly sedated. It is important to note that not every follicle will contain an egg, and not every egg will fertilise.
The key difference between ICSI and IVF is the insemination process. In ICSI, a single sperm is directly injected into a mature egg by a skilled embryologist, whereas in IVF, multiple sperm are added to the eggs and left to fertilise naturally without an embryologist’s intervention.
On the day of egg collection, the semen sample is concentrated, and each mature egg is injected with a selected sperm. The sperm is chosen based on its normal morphology and binding to the media, indicating better DNA integrity for higher embryo development success. Fertilisation is assessed 16-18 hours after injection.
The inseminated eggs are placed in an incubator and checked for fertilisation 16-18 hours after insemination. You will receive an update from the embryologists at this time. The normally fertilised embryos are then grown for five days.
Whilst every procedure is designed to provide the best chance of fertilisation and optimal embryo development, it is important to note that not all eggs will fertilise, and not all fertilised eggs will develop into embryos.
Five days following egg retrieval, the embryos are assessed by the embryologists for development and quality. The best embryo is selected for transfer into the uterus using a fine plastic tube (transfer catheter). Usually, only one embryo is transferred. If more than one embryo is to be transferred, this must be discussed with your clinician prior to the transfer. Embryo transfer is a relatively quick procedure, and discomfort should be minimal.
In addition to fresh embryo transfers, many patients may have embryos frozen for future use. This process is known as a Frozen Embryo Transfer (FET), where a cryopreserved embryo from a previous cycle is thawed and placed into the uterus, which is widely used in IVF treatment. FETs avoid the need for another full IVF cycle, reducing treatment time, costs, and physical demands. Learn more about how FET works and if it may be right for you on our Frozen Embryo Transfer (FET) service page.
Any remaining embryos that meet the development and quality requirements will be frozen. These frozen embryos are then stored until required for a subsequent transfer. The vast majority of frozen embryos survive the freezing process, and pregnancy rates are comparable with the transfer of fresh embryos.
The final stage of ICSI IVF is called the luteal phase, and it’s the two weeks between the egg collection and the pregnancy test. Many people find this to be the most difficult time to endure. Whilst it’s an exciting and optimistic time of the treatment cycle, it can often be emotionally charged with expectation and anxiety. You will be encouraged to limit your activity for 24 hours after the embryo transfer, eat well, stay hydrated and keep busy while you wait for your pregnancy blood test.
To learn more about the ICSI procedure, please read our fact sheet: ICSI or visit our Patient information booklets page. You can also view our ICSI PDF to find out more about the process.
Who is ICSI recommended for?
ICSI is commonly recommended in cases of male factor infertility. It can assist when sperm quality or function makes natural fertilisation difficult, including situations such as:
- Abnormally shaped sperm.
- Slow moving sperm.
- A low sperm count.
- An obstruction such as a vasectomy, preventing sperm release.
- Antisperm antibodies (those produced by the man’s body and may inhibit sperm function).
- A vasectomy reversal that was unsuccessful and resulted in a low sperm count or poor-quality sperm.
ICSI may also be considered for couples who have previously undergone IVF with reduced or no fertilisation.
Frequently Asked Questions
ICSI is an additional cost on top of a standard IVF cycle because it involves a specialised laboratory procedure where a single sperm is injected directly into each egg, requiring extra time, skill and resources from the scientific team.
The overall cost of treatment can vary depending on several factors, including medications, additional procedures such as PICSI, and genetic testing. Some components of fertility treatment may attract Medicare rebates, and private health insurance may contribute towards hospital-related costs, depending on your level of cover. Payment plan options are also available to help manage treatment expenses.
For a detailed breakdown, please visit our pricing page.
At City Fertility, around 70% of mature eggs will fertilise normally after ICSI. However, it is important to understand that fertilisation rates can vary depending on several individual factors, including egg and sperm quality, age, and overall health.
While ICSI is a highly effective technique for overcoming male factor infertility, fertilisation does not guarantee pregnancy. Not all fertilised eggs will develop into healthy embryos, and not all embryos will implant successfully. Your fertility specialist will discuss your individual circumstances and the factors most relevant to your chances of success.
ICSI involves injecting a single sperm into a single mature egg. Standard IVF involves adding many sperm to a dish containing the eggs. The sperm and eggs are left for a period of time to allow the sperm to penetrate the egg naturally. ICSI is recommended for couples with fertility challenges due to male factors or those where standard IVF has been unsuccessful.
Conventional ICSI selects sperm based on appearance (morphology only). The basis for this is that sperm with a normal appearance are less likely to contain damaged DNA. Physiological Intracytoplasmic Sperm Injection (PICSI) uses additional factors to select the sperm. At City Fertility, we use SpermSlowTM to perform PICSI. Sperm that are bound by the medium are less likely to have damaged DNA, thus giving the embryologist extra assistance in selecting better quality sperm. To learn more, view our PICSI PDF.
ICSI is usually recommended when male factor infertility is present, such as low sperm count, slow-moving sperm, abnormally shaped sperm, or antisperm antibodies. It may also be used in cases where a vasectomy reversal has been unsuccessful and resulted in a low sperm count or poor-quality sperm.
ICSI may also be considered for couples who have previously undergone IVF with reduced or no fertilisation.
The ICSI process fits within a standard IVF cycle. Ovarian stimulation starts on Day 2 or 3 of the menstrual cycle, followed by an hCG “trigger injection” and egg collection 35–38 hours later. On the same day, each mature egg is injected with a single sperm, and fertilisation is checked 16–18 hours after injection. Embryos are cultured for up to five days, with the best embryo usually transferred on Day 5. Any remaining suitable embryos may be frozen for future use.
No. The chance of twins or multiple pregnancies depends on the number of embryos transferred, not the ICSI procedure itself. Usually, only one embryo is transferred, and if more than one is being considered, this will be discussed with your clinician.
ICSI involves more handling of the egg, so there is a higher risk of egg damage than in standard IVF. In addition, some male infertility factors may be passed on to children. For example, male children may experience similar fertility challenges to their father, and in some cases, there is an increased risk of inheriting genetic conditions such as cystic fibrosis or sex chromosome abnormalities.
ICSI carries a higher risk of damage compared to standard IVF procedures. Increased handling of the oocyte (egg) carries a higher risk of damage compared to standard IVF procedures.
For the child:
- Male children born from ICSI may experience the same fertility challenges as their father (depending on the cause)
- Some children born to men with very low sperm counts have an increased risk of inheriting genetic abnormalities such as cystic fibrosis or sex chromosome abnormalities. These are inherited conditions and may be screened for prior to treatment. If these conditions are confirmed, your fertility specialist will discuss your options, which may include testing of embryos.
Please remember that anyone can be affected by infertility, and while it can be a challenging experience, the good news is that our fertility specialists can help you explore the options available to achieve your dream of having a baby.